FREE SALE LICENSE
Certificate of Free Sale (Free Sale License)
A free sale license, also known as a certificate of free sale, is a document that certifies that a particular product is legally sold in the country where it is manufactured and is considered safe for public consumption. The purpose of this license is to allow manufacturers and exporters to prove to foreign regulatory authorities that their products are in compliance with local laws and regulations.
A free sale license is typically issued by the government of the country where the product is manufactured. The license may be required by foreign countries as part of the import process for certain types of products, such as pharmaceuticals, medical devices, or food and beverage items. The license serves as proof that the product has been inspected and tested and meets all safety and quality standards.
Obtaining a free sale license may involve submitting an application to the appropriate government agency, providing documentation such as product testing reports and safety certifications, and undergoing an inspection or audit of the manufacturing facility.
In summary, a free sale license is a certificate that attests to the safety and legality of a product for sale in the country of origin, which can be used as evidence to facilitate international trade.
BENEFITS TO USERS
Cost Savings: Users can access and use the product or service without having to pay for it, saving them money.
Try Before Buying: Users can test out the product or service without any financial commitment, allowing them to make an informed decision before making a purchase.
Accessibility: A free sale license opens up the opportunity for individuals or organizations with limited budgets to access and benefit from the offering.
Exploration and Learning: Users can explore and learn about the features and benefits of the product or service, helping them understand its value and potential.
Flexibility: With a free sale license, users have the freedom to use the product or service as they wish, without any restrictions or limitations.
What are the chargeable fees of a Free Sale License?
The government fees for obtaining a free sale license certificate in India may vary depending on the type of product and the regulatory authority that issues the certificate.
For example, the Central Drugs Standard Control Organization (CDSCO) in India charges a government fee of INR 5,000 for a free sale license for drugs and medical devices, while the Food Safety and Standards Authority of India (FSSAI) charges a government fee of INR 5,000 for a free sale license for food and dietary supplements.
It is important to note that the government fees for obtaining a free sale license may also be subject to change, and it is advisable to check with the relevant regulatory authority for the most up-to-date fee information. Additionally, there may be additional costs associated with the application process, such as fees for inspections, testing, and documentation, which may vary depending on the product and the regulatory requirements.
VALIDITY OF FREE SALE LICENSE
The validity of a free sale license typically depends on the country where it was issued and the type of product it pertains to. In most cases, a free sale license is valid for a specific period of time, usually ranging from one to five years, after which it may need to be renewed or reissued.
It is important to note that a free sale license only confirms that the product is legally sold in the country of origin and complies with local regulations. It does not guarantee that the product will meet the regulations of other countries or that it will be accepted by foreign regulatory authorities. Importing countries may have their own regulatory requirements and may conduct their own inspections or testing of the product before allowing it to be sold.
Therefore, even if a free sale license is valid and up-to-date, exporters and manufacturers may need to comply with additional requirements and obtain other certifications or licenses in order to export their products to foreign markets.
Which products need a certificate of free license in India ?
A certificate of free sale, or free sale license, may be required for a variety of products, depending on the laws and regulations of the country of origin and the importing country.
In India, the products that typically require a certificate of free sale or free sale license include:
- Pharmaceuticals and medical devices: All drugs, medicines, and medical devices require a free sale license from the Central Drugs Standard Control Organization (CDSCO) to ensure their safety and efficacy.
- Food and beverages: Certain food products such as dietary supplements, infant food, and fortified food products require a free sale license from the Food Safety and Standards Authority of India (FSSAI) to ensure their safety and compliance with food safety regulations.
- Cosmetics: Certain cosmetic products such as skin lightening creams, hair dyes, and other cosmetic products require a free sale license from the CDSCO to ensure their safety and compliance with cosmetic regulations.
- Animal products: Animal products such as pet food and livestock feed require a free sale license from the Department of Animal Husbandry and Dairying to ensure their safety and compliance with animal welfare regulations.
- Agricultural products: Certain agricultural products such as seeds, pesticides, and fertilizers require a free sale license from the Ministry of Agriculture and Farmers Welfare to ensure their safety and compliance with environmental regulations.
It is important to note that the specific products that require a free sale license in India may vary depending on the regulatory environment and the relevant regulatory bodies. It is advisable to check with the appropriate authorities to determine if a free sale license is required for a particular product.
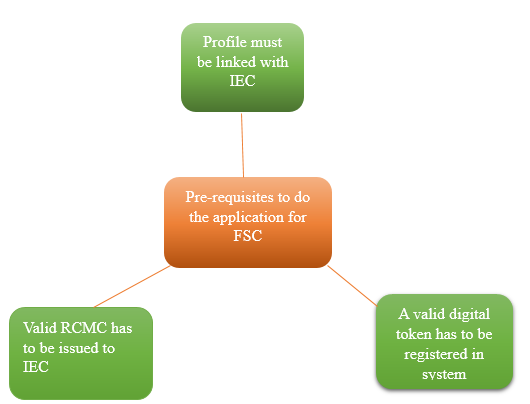
Prerequisite for the application to get a Free Sale Certificate
For the Free Sale and Commerce Certificate following requirements are there before proceeding with the application
Profile on DGFT portal has to be linked with active Import – Export Code.
A valid Digital token has to be created in the system; the same can be verified from My Dashboard.
An RCMC has to be issued to the IEC.
Compliance in free sale license in India?
In India, compliance in free sale license refers to the requirement that the product for which the free sale license is being obtained must comply with all applicable regulations and standards set forth by the relevant regulatory authority. The specific requirements for compliance may vary depending on the type of product being sold and the regulatory authority responsible for overseeing its safety and quality.
For example, in the case of drugs and medical devices, the Central Drugs Standard Control Organization (CDSCO) is the regulatory authority responsible for overseeing the safety and efficacy of these products. Compliance with CDSCO regulations involves meeting requirements for product quality, safety, efficacy, and labelling, as well as conforming to good manufacturing practices (GMP) and other relevant standards.
Similarly, the Food Safety and Standards Authority of India (FSSAI) is responsible for ensuring the safety and quality of food products sold in India. Compliance with FSSAI regulations involves meeting requirements for food safety, hygiene, labelling, and packaging, as well as conforming to relevant standards such as the Food Safety and Standards Act, 2006 and the Food Safety and Standards (Packaging and Labelling) Regulations, 2011.
Non-compliance with the regulations and standards set forth by the regulatory authority may result in the revocation of the free sale license, fines or other penalties, and in severe cases, legal action against the manufacturer or distributor of the product. Therefore, it is important for companies to maintain ongoing compliance with all relevant regulations and standards in order to ensure the continued validity of their free sale license in India.



Leave a Reply